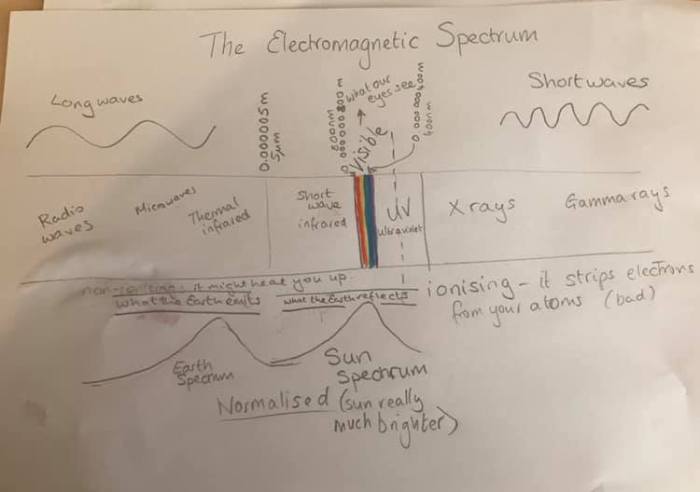
To understand climate change, we first need to understand light. (Personal aside: I got a prism for my tenth birthday and told everyone that one day I’d get a job splitting light into pretty colours – so of course I start here)
Light is electromagnetic waves that travel at the “speed of light”. The properties of light depend on the wavelength (how many times the electromagnetic field vibrates). Short wavelength light vibrates lots and the wavelength is small enough to get inside you and damage you – that’s “ionising radiation”: ultraviolet that damages your skin and x-rays and gamma rays that go inside.
Long wavelength light is radio and microwaves and the infrared. That can’t damage your molecules directly, but (and we’ll come back to this), some infrared and microwaves can make molecules vibrate which heats things up.
In the middle is visible light – the bit we can see. At 400 nm (nanometre – that means 0.000 000 400 m) wavelength we start to see blue light (if we don’t have cataracts) around 555 nm it looks quite green – and our eyes are most sensitive. At 800 nm we just about see a deep red (unless colour-blind and lacking red sensors).
Now it’s no coincidence that this is the bit of the electromagnetic spectrum that we see best. This is the peak of the sun’s spectrum – and all we see on Earth is visible electromagnetic radiation from the sun (or one of our artificial lights) that reflects from the Earth.
But the Earth itself does glow – just in wavelengths we don’t see. We call that the thermal infrared. In lesson two I’ll explain about blackbody radiation.
2 thoughts on “Lesson 1: Electromagnetic Radiation”