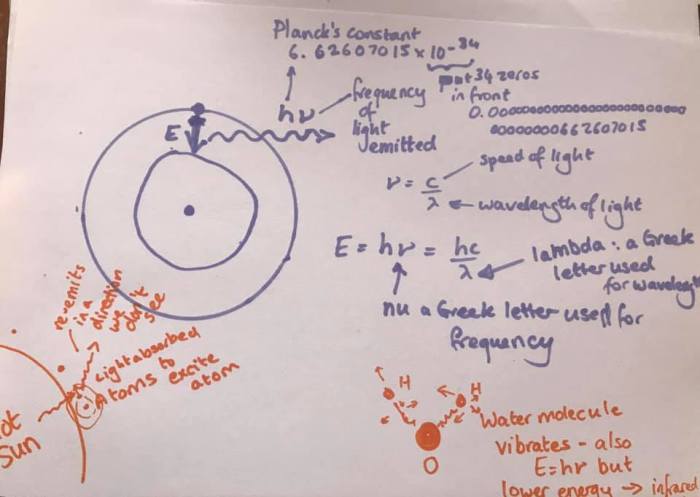
When I was 17 and doing A’level chemistry I learnt the equation E = h nu. This was the most exciting science lesson of my life. Seriously. I was only disappointed that chemistry rather than physics gave me that gift 😂
You see, I’ve always been fascinated by colour – the mixture of a prism for my tenth birthday and my dad being colourblind (a condition one of my two sons has inherited from the x-chromosome I got from my dad: the other son got the x-chromosome I got from my mum) meant that I really wanted to understand how “colour worked”. Now there are three bits to that – understanding our eyes (a detour I won’t go down now), understanding blackbodies – which explains white light – where lots of wavelengths are present, which we covered before, and understanding E = h nu which explains lights being coloured in and of themselves.
What this equation shows is how atoms interact with light. You’ll remember the simplified model of an atom with a central core and electrons in rings around it? Well, when an atom is “excited” the electrons can go up to a higher orbital. And when they fall back down to the ground state (the state where all the electrons are as close to the nucleus as possible while obeying the rules of how many electrons can be in each orbital) they release energy (based on the size of the jump) as light and the frequency of the light (nu) is proportional to the energy jump. So the bigger the energy of the jump, the higher the frequency of the light (more blue, less red).
You can see this yourself. Drop salt into a flame and the heat of the flame will excite the sodium atoms in salt to a higher energy state. As they fall back down they release yellow light. You might recognise that yellow light if you remember sodium street lights. In those electricity excited the sodium atoms. (Go on, try it!)
In the sun’s spectrum there are black lines at special wavelengths (called Fraunhofer lines). That is this process in reverse. The outer part of the sun is cooler than the inner part and absorbs light to make electrons jump up to higher orbitals. So the blackbody light from inside the sun (all wavelengths) loses light at the special E = h nu wavelengths. Of course those do re-emit those wavelengths as they drop back down, but here’s the crux: they re-emit in all directions including back towards the middle, so the amount of light coming towards us is lower.
This is how helium was discovered – they could match most Fraunhofer lines to lines they could get by putting elements into flames – but there were a set of lines they couldn’t match, so they proposed a new element – helium. (From Helios). Later they found helium on Earth. This is also how neon lights work – different jumps in neon give the different colours of neon light.
Now atomic lines are high energy so they tend to be in visible spectral bands. Later we’ll see that molecules have absorption/emission lines too. These are not from electrons jumping but from the molecule wobbling. Because whole atoms have to move the energy is much lower (heavier things don’t move as easily). So these lines are in the infrared.
But we’ll come back to that.
One thought on “Lesson 3: My favourite equation”