
I showed the picture above in the previous lesson and discussed how water vapour absorbs a very broad set of wavelengths in the thermal infrared (and a few in the near infrared). This absorption is due to how the light of those wavelengths causes the water molecules to change their vibrational modes in lots and lots of different ways.
The carbon dioxide molecule has three atoms arranged in a straight line: a carbon atom in the middle and two oxygen atoms either side. It doesn’t have quite as many ways of vibrating as water, but it has quite a few – and crucially different ones (pink in the diagram above), so it absorbs thermal infrared at wavelengths that water vapour cannot respond to. Thus, carbon dioxide removes even more wavelengths that the Earth can use to cool down in outgoing radiation.
In the last lesson, I also described the water feedback loops – simplistically if there’s too much water vapour in the atmosphere it rains. More completely, a higher temperature means both more water vapour in the atmosphere as hot air holds more water – creating more heating – and it means more clouds which may either accelerate warming (trapping heat in at night), or slow down warming (reflecting more sunlight in the day time) – but we’re not quite sure which.
We are increasing the amount of carbon dioxide in the atmosphere (we’ll come back to the evidence for that later – but basically, for most of the last ten thousand years there were 250-280 carbon dioxide molecules in a million air molecules and now there are 400). And there isn’t a feedback loop as simple and immediate as “rain” to get rid of it. There are ways it can naturally come out of the atmosphere: the main ones are increased plant growth (eg in rainforests) and increased ocean algae. The oceans can also absorb some carbon dioxide, but that makes them more acidic which impacts marine life – particularly corals. Of course, if we’ve cut down the rainforests (which we really have) they can’t absorb as much carbon dioxide either.
Because I’m still taking about the basic physics, I want first to consider what the increased carbon dioxide (wherever it comes from) does.
Now you might think that’s easy – CO2 is a greenhouse gas so more CO2 means more warming; but that isn’t directly true. The atmosphere is very thick – so the thermal infrared meets lots of carbon dioxide molecules on the way up: that means that the atmosphere already absorbs all the light at some wavelengths (the ones where the graph above touches the top of the image). Increasing the concentration of carbon dioxide might make it be fully absorbed slightly earlier, but you can’t be more absorbed than fully absorbed (and at some wavelengths it only takes 25 metres of air to block the light completely).
Instead there are two important effects. The easier effect to understand is that not all infrared wavelengths are completely blocked by the atmosphere. In the last lesson I showed a graph of atmospheric absorption zoomed in and there you see lots and lots of thin lines. As the concentration of carbon dioxide in the atmosphere increases, some of those lines get broader, and some of them get deeper. For example, some wavelengths represent changes from an unusual vibrational mode to another, that are rarely “set up” – it’s rare for the light to meet a molecule in the right starting state, but when there are more carbon dioxide molecules, the light is more likely to find one of these rare molecule vibrational states, so those wavelengths are more frequently absorbed and the absorption line deepens.
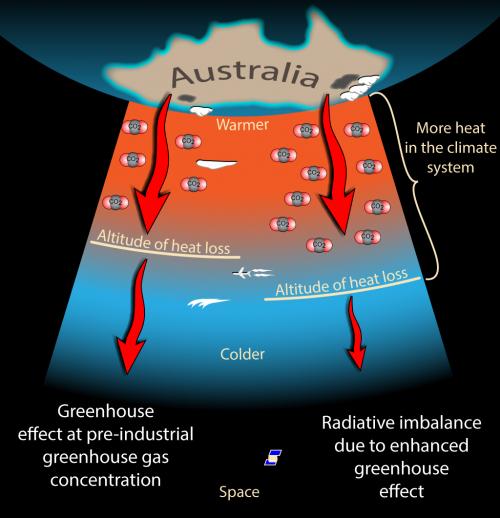
The more subtle effect is that the atmosphere itself is also lots of little blackbodies radiating thermal infrared blackbody spectra that depends on the temperature of the gases. (As the thermal infrared radiation is absorbed by the carbon dioxide and water vapour in the atmosphere it heats the atmosphere up).
At low altitudes, any infrared emitted by the atmosphere is absorbed by the carbon dioxide molecules and can’t make it through. But there is a height where the atmosphere can radiate to space because there aren’t enough carbon dioxide molecules above it. Increasing the concentration of carbon dioxide means more molecules throughout the atmosphere and therefore this level has to go up towards space (at the lower height where light once could escape it now is more likely to hit other molecules and therefore not escape). Since the higher parts of the atmosphere are colder, there is less energy escaping to space than would be there at lower levels (smaller blackbody curve at lower temperatures) – so the planet loses less heat.
Eek. Sorry. I could have over-simplified this: more CO2 means more greenhouse warming. But I want to try to explain the whole story as I understand it (I am not an expert on climate modelling, so there are still huge simplifications in here I don’t know about!)
One last point: water vapour and carbon dioxide are not the only greenhouse gases. Methane is another important one – with four hydrogen atoms round a carbon atom, it has a lot of vibrational modes – but there’s not as much of it in the atmosphere as there is carbon dioxide. It is also increasing. The refrigerants (HFCs, HCFCs, CFCs) don’t only damage the ozone layer (ozone has vibrational modes that block UV on the way in) but are also very potent greenhouse gases – partly because they don’t occur naturally so they absorb wavelengths nothing is absorbing already. There are currently very low levels of these, but if we don’t dispose of our old refrigerators and air conditioning units carefully we’ll release them into the atmosphere and because there is no absorption at these wavelengths already – a small increase makes a big difference. Just think about how many air conditioning units there are – and the human feedback loop: more warming, more air conditioning, more refrigerant gases, more warming… (that’s why Project Drawdown puts disposing of refrigerant gases carefully as their number 1 activity for solving climate change problems).
One thought on “Lesson 7: Carbon dioxide as a greenhouse gas”