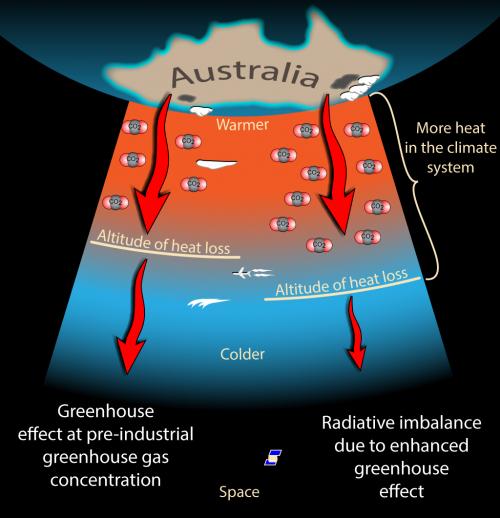
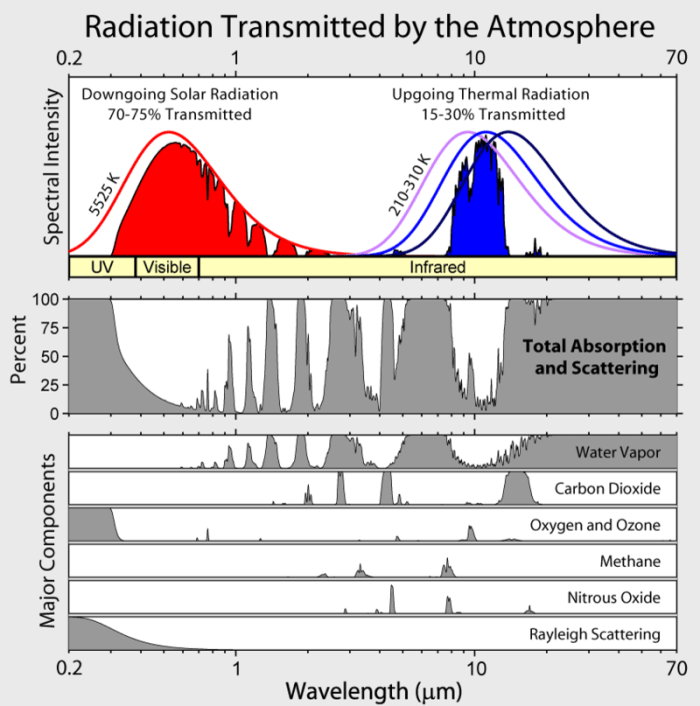
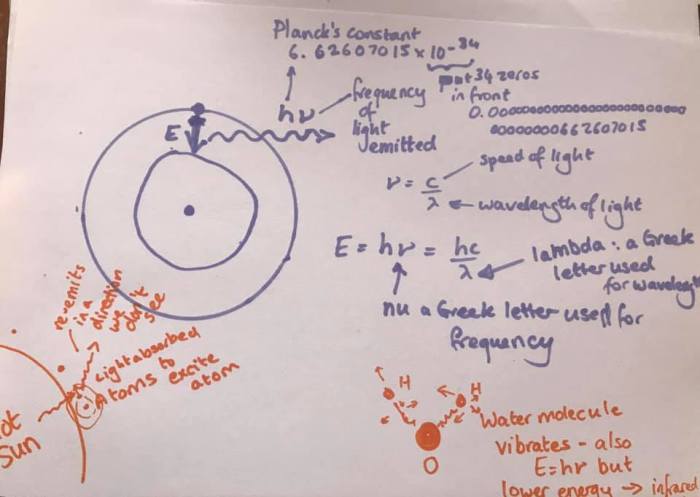
So, we’ve discussed blackbody radiation and how the hot sun emits electromagnetic radiation at short wavelengths (UV, Visible, near IR) and the much cooler Earth radiates in the thermal IR. We’ve discussed how the Earth needs to reach an equilibrium where the incoming energy matches the outgoing energy and how without greenhouse gases that would be achieved at around -18 ºC, but, because greenhouse gases absorb thermal IR to excite various vibrational modes (make the molecules wobble), a lot of the thermal IR gets absorbed in the atmosphere and the Earth warms up.
I hope I’ve expressed two core concepts: these processes are all basic physics and chemistry in and of themselves, but there is complexity in the Earth system because of interactions and feedback loops. It’s not quite as simple as more CO2 means more vibrating molecules and hence more warming: increasing CO2 does cause warming, but to understand how much, you need to understand exactly how the light interacts with all the molecules and how the atmosphere itself radiates and how increasing atmospheric temperature holds more water vapour which also acts as a greenhouse gas. It’s both very simple – and very complicated!
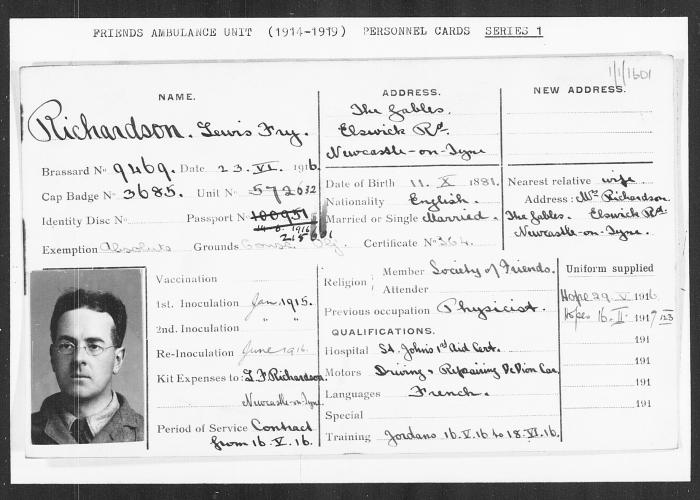
Now, a slight aside to get to how that complexity is handled. Back in World War 1 a young Quaker (this is a subject that brings together both my faith and my science!), Lewis Fry Richardson was working in the Friends’ Ambulance Unit in the trenches. By day he dealt with the wounded and the dying. And at night he solved differential equations. I get that: after the horrors of the day, maths provided the rational logic that helped him control emotions.
What he was trying to do was to make the first weather forecast. He had weather measurement data for an area in Central Europe and he decided he’d try to predict the temperature in one place by using what had happened six hours earlier in other places. He had the concept of the first numerical weather forecast. The idea was simple; he would split his map up into lots of different cells and then in each cell he would know both the current temperature, pressure, wind speed and direction and, crucially, how that was changing with time (what in maths is known as “the differential”). He’d solve the differential equations in each cell and that would pass information to the next cell. That way he could calculate numerically what the weather would be a six hours later in one of his cells. He spent six weeks on his calculations – and ended up with the wrong answer (I know that feeling too!). We now know that his wrong answer was because of problems with the input data (the measurements of temperature and pressure that he had were not reliable enough – we’ll certainly come back to that message since my job is to make sure the measurements that go into models are reliable!)
However, his principle was right – you can predict the weather in one place by cutting the Earth up into lots of cells, using measurements and estimates of the current conditions in each place and the rate of change of those conditions, and then solving numerically the differential equations in each cell to show the change until the next time period. He knew that it had taken him 6 weeks to calculate the one cell he was working on, but he imagined that if there were 64000 (human) calculators working together, they could do real time weather forecasting and predict the future. His concept of a “weather forecast factory” (illustrated above) and is exactly what is done in the supercomputers that run today’s weather forecasts.
We’ll go into them in more detail in a later lesson, but basically numerical weather forecast models split the Earth and its atmosphere and oceans into lots of “cells” – boxes that cover a certain longitude and latitude at a particular atmospheric height (or ocean depth). In each box they model the basic physics of radiation (heat, light, temperature) and convection (air/water pressure and winds/water currents) in each box and solve differential equations to show how that is changing over a defined time step. Modern models also model the chemistry (how gases in the atmosphere interact with each, changing salinity and pH of the oceans) and biology (growth of plants and algae, respiration) as well as the large scale geoscience (sun irradiance changes, volcanoes, …).
Numerical weather forecasts are some of the most complex computer programs in the world, being run on some of the biggest and most powerful computers in the world.
The “short term weather forecast” models (which can accurately predict ~3-5 days), the “medium term weather forecasts” and the “climate forecasts” all run exactly the same model at the UK MetOffice – they just use smaller cells and do the calculation on a much finer time scale for weather forecasting and use bigger cells and averages over a month on the climate forecasting. Each meteorological office has its own model developed by its own scientists and programmers – and even within one meteorological office they may have multiple variations of their model. That’s how they can say “there’s a 70% chance of rain” – what they mean is that when they ran their model many times with minor changes to account for what they didn’t know, 70% of the models put out rain and 30% didn’t.
Now I know what you’re thinking! If you’re British and older than 40 you’re remembering Michael Fish on the BBC saying there wouldn’t be a hurricane the day before the 1987 storm. I remember that day vividly as I tried to cycle to school around the fallen trees and got there to find school was closed – which is sort of the point – I couldn’t check in advance if school was closed because there was no (well no established) internet: computers were significantly less powerful back then. The weather forecasts of today are much more sophisticated and much more accurate. But, granted, they are only accurate for around 3-5 days (and we all know there is a limit – the famous “butterfly effect” that means minor changes make big differences to a chaotic system – so we can’t predict more than about 10 days ahead, no matter how sophisticated our models and how powerful our supercomputers).
So how can we predict climate with the same models? The reason for that is that with climate we’re asking a somewhat different question – instead of asking “what will the temperature be at Heathrow at 10 am on the 3 June 2080?” we’re asking “what will the average temperature be for all Junes in the 2080s in outer London?” That’s a different question – and ones the models, with bigger cells and more time averaging, can answer.